US Quiz of the Month – January 2022
CASE REPORT
The authors present the case of a 72-year-old male patient being studied at Gastroenterology Consult due to hyperbilirubinemia (Total bilirubin 2mg/dL). Gilbert syndrome and toxic, metabolic, virologic and auto-imune causes were excluded. Abdominal ultrasound showed gallbladder lithiasis. Abdominal magnetic resonance disclosed a pericentimetric periampullary nodularity.
An endoscopic ultrasonography (EUS) was performed and identified a hyperechoic thickening of the ampulla, with clearly defined regular borders, without intraductal infiltration, biliary or pancreatic duct dilation (Fig. 1).
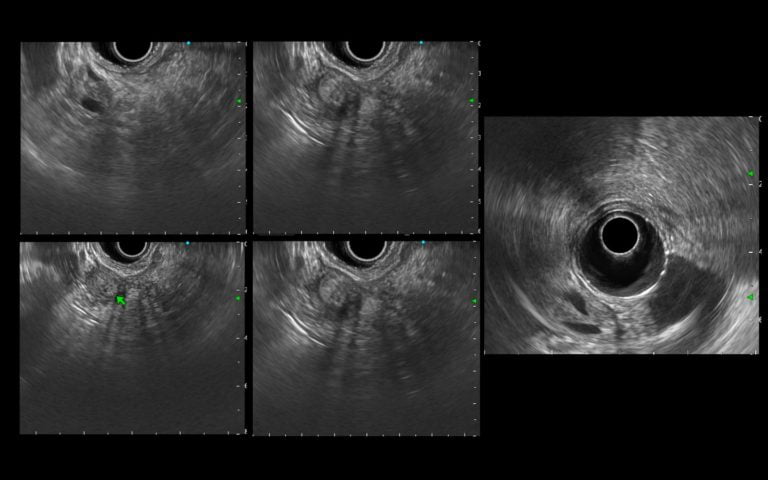
Figure 1. EUS (transduodenal view): A heterogeneous hyperechoic thickening of the ampulla, with a clearly defined regular border, without intraductal infiltration, biliary or pancreatic duct dilation.
WHAT IS THE MOST LIKELY DIAGNOSIS?
DISCUSSION
These findings were compatible with ampullary adenoma. Duodenoscopy showed a papilla with a polypoid appearance, with around 15mm, without ulceration. EUS evaluation showed signs of benign nature: the duodenal layers were preserved, there were no biliary or pancreatic duct dilatations and there was no evidence of intraductal polypoid infiltration. Multiple biopsies of the papilla of Vater were performed, that disclosed adenoma with low-grade dysplasia. The patient accepted and an endoscopic papillectomy was performed. Histologic assessment confirmed the adenomatous nature, with low-grade dysplasia. There were no signs of submucosal infiltration and resection margins were clear. Distal biliary duct biopsies were performed during the same session: bile duct type mucosa with fibrosis, without dysplasia or neoplastic tissue. Total bilirubin has normalized, and the patient is asymptomatic. Follow-up at three months with duodenoscopy and biopsies showed no signs of endoscopic or histologic recurrence.
Nowadays, ampullary tumors are being increasingly diagnosed, frequently during routine upper endoscopy performed for unrelated reasons [1,2]. The majority of benign and malignant ampullary tumors are sporadic and involve the major papilla; however, they can also occur more frequently in the setting of polyposis syndromes. The adenoma can arise from intestinal-type mucosa or pancreatic duct-type ampullary mucosa [3]. They are considered premalignant, and the adenoma-carcinoma sequence has been assumed to be behind the pathogenesis of ampullary cancer like other locations within the GI tract[2]. On EUS the adenoma is visualized as a hypoechoic or hyperechoic and homogenous thickening of the papilla, without invasion of the duodenal wall. If the view is optimal, it is possible to recognize harmless submucosal and duodenal muscularis propria layers [2]. The sensitivity and specificity of EUS in the detection of T1 tumors are reported to be 77% and 78%, respectively [4]. Malignant features of an ampullary tumor include a hypoechoic mass with irregular margins and areas invading the surrounding tissue. Invasion can be seen as a continuation of the main hypoechoic mass into adjacent structures [5]. Other malignant findings are the presence of peri-duodenal adenopathy, bile or pancreatic duct dilatation or intraductal growth [2].
When an ampullary tumor is suspected, a side-viewing endoscopy and biopsies must be performed, as well as a EUS or a magnetic resonance cholangiopancreatography, to access the preoperative staging. If biopsies confirm an ampullary adenoma, attention must be taken if there is an intraductal extension >20mm. Endoscopic papillectomy is indicated when there is an ampullary adenoma with low- or high-grade dysplasia, with ≤ 20mm intraductal extension, and a size between 20 and 30mm. The management of adenomas between 30-40mm must be considered on a case-by-case basis. When complete resection is achieved (R0) after endoscopic papillectomy, follow-up must be made at three months with duodenoscopy and biopsies. On the other side, if the biopsies disclose an ampullary carcinoma or an adenoma with intraductal extension >20mm or if endoscopic papillectomy is technically difficult (size >4cm, diverticulum), a surgical option must be discussed [1].
REFERENCES
- Vanbiervliet G, Strijker M, Arvanitakis M, Aelvoet A, Arnelo U, Beyna T, et al. Endoscopic management of ampullary tumors: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2021;53(4):429–48.
- Hawes RH, Fockens P, Varadarajulu S. Endosonography, Fourth Edition, (2019) [Internet]. Endosonography. 2019. 1-14.e1 p. Available from: www.elsevier.com/permissions.
- Fischer H-P, Zhou H. Pathogenesis of carcinoma of the papilla of Vater. J Hepatobiliary Pancreat Surg. 2004;11(5):301–9.
- Trikudanathan G, Njei B, Attam R, Arain M, Shaukat A. Staging accuracy of ampullary tumors by endoscopic ultrasound: meta-analysis and systematic review. Dig Endosc Off J Japan Gastroenterol Endosc Soc. 2014 Sep;26(5):617–26.
- Lim GJ, Devereaux BM. EUS in the assessment of ampullary lesions prior to endoscopic resection. Tech Gastrointest Endosc [Internet]. 2010;12(1):49–52. Available from: https://www.sciencedirect.com/science/article/pii/S1096288310000094
AUTHORS
Carolina Chálim Rebelo1, Nuno Nunes1, Diogo Bernardo Moura1, José Renato Pereira1, Maria Antónia Duarte1.
- Gastroenterology Department, Hospital do Divino Espírito Santo de Ponta Delgada, Ponta Delgada, Portugal.


