US Quiz of the Month – March 2020
CASE REPORT
We present a case of a 52-year-old patient with acromegaly and pituitary macroadenoma, previously resected and treated with adjuvant radiotherapy due to remnant adenoma, referred to our unit due to a large multiloculated cystic lesion of the pancreatic tail diagnosed during follow-up imaging. The patient was asymptomatic and had no previous medical history of abdominal complaints. Abdominal MRI reported several giant hepatic hemangiomas and a pancreatic tail multiloculated cyst, with 80 x 58 mm, with thick septa but no mural nodules (Fig. 1).
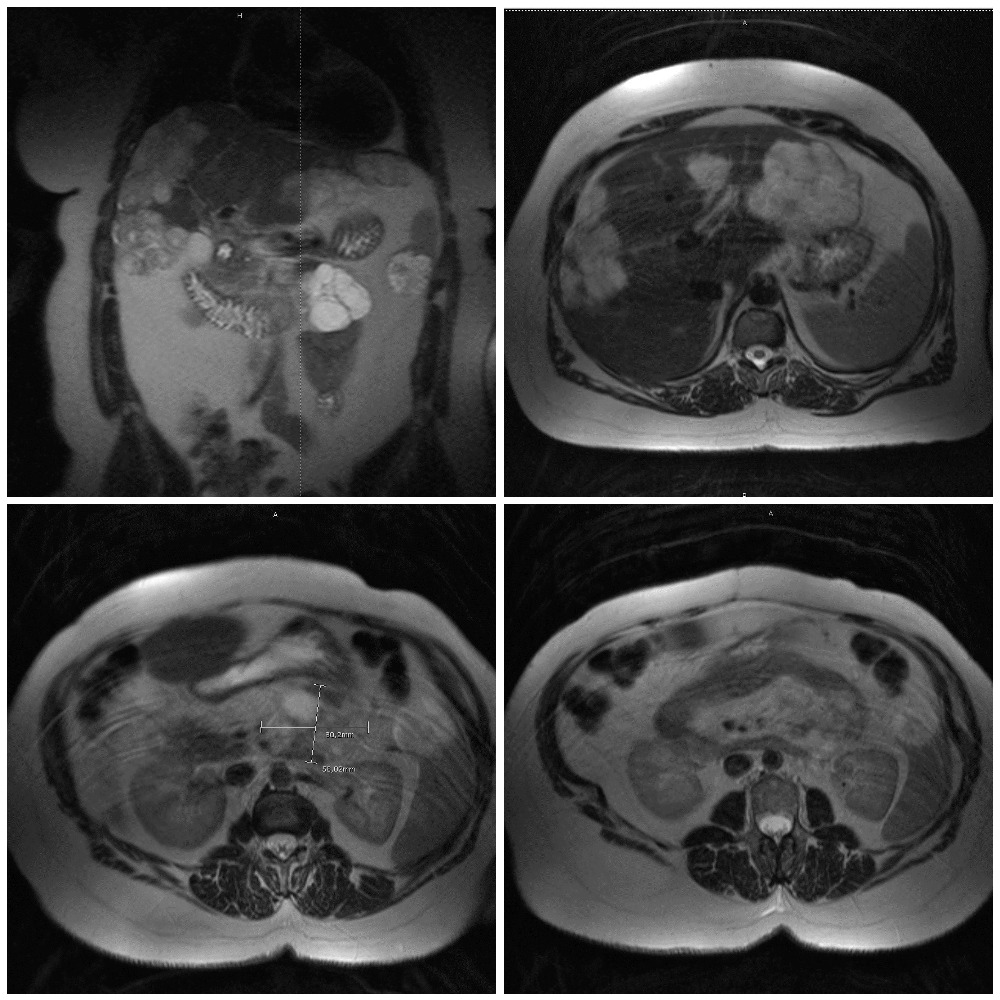
Figure 1. MRI (axial view): giant hepatic hemangioma; multiloculated cyst with 80 x 58 mm, with thick septa in the pancreatic tail.
Tumor markers were within normal range. EUS was performed and a large macrocystic lesion was identified in the pancreatic tail, mostly with anechoic content, with 64 x 50mm and without wall thickness or mural nodules. Pancreatic duct was within the expected diameter and apparently showed no communication with the lesion (Fig. 2). An EUS-FNA with a 19-gauge needle was performed and a chylous-like cystic fluid was obtained (negative string sign) (Fig. 3).
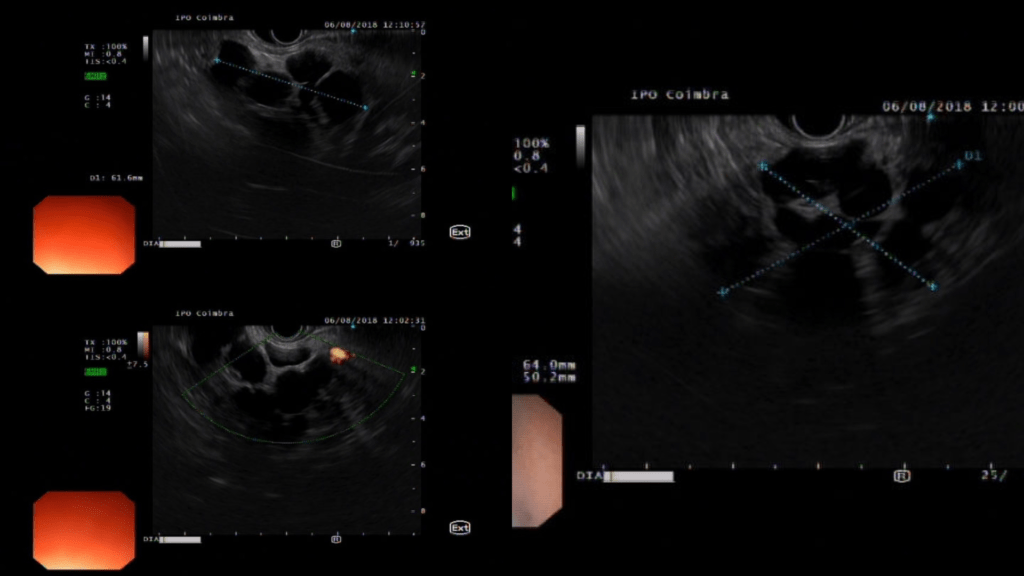
Figure 2. EUS: Multiloculated cyst, anechoic content, no mural nodules.
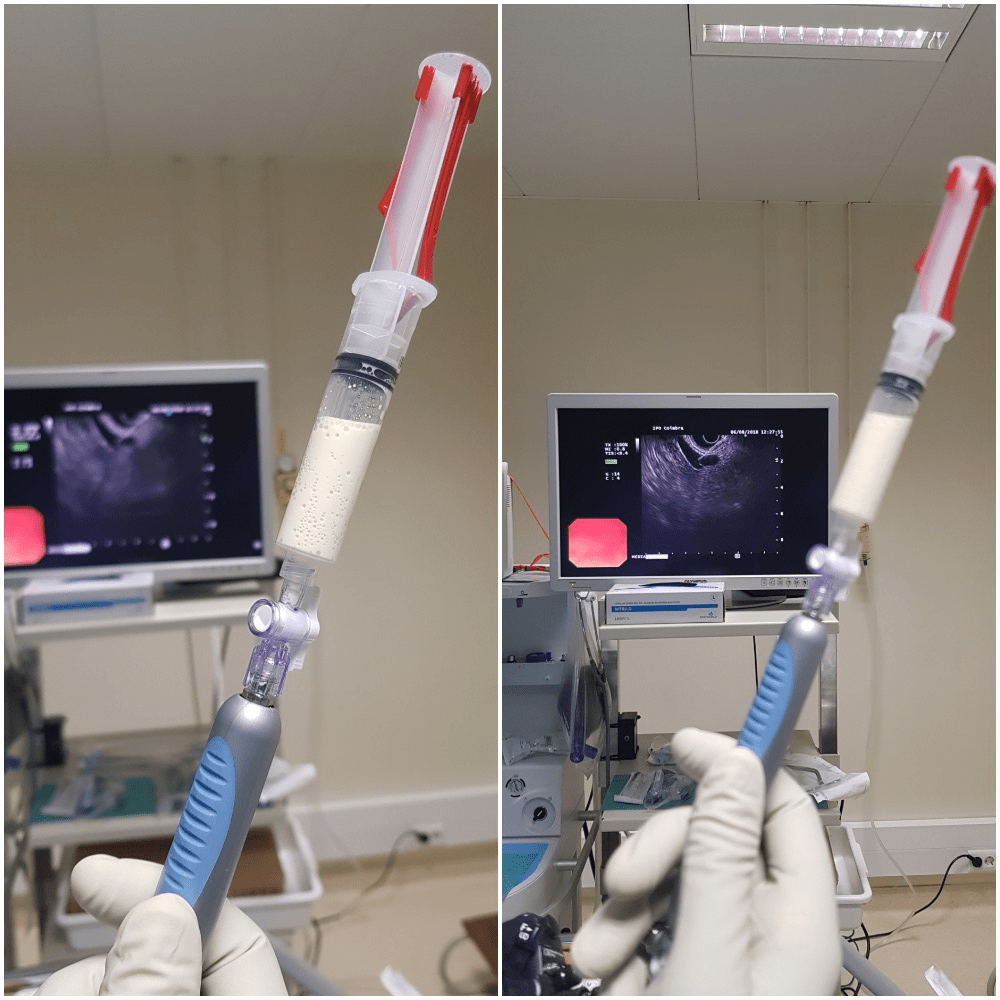
Figure 3: EUS-FNA: Aspirated whitish and chylous-like liquid.
WHAT IS THE MOST LIKELY DIAGNOSIS?
DISCUSSION
Biochemical evaluation of the cyst liquid revealed amylase and CEA within the normal range and a high triglycerides count – 1028 mg/dl. Cytology was suggestive of non-mucinous pancreatic cyst. The diagnosis of pancreatic lymphangioma was then made. This case was presented at the tumor board with no indication for surgery.
Lymphangiomas are rare benign, slow-growing cystic lesions, with a female predominance. Less than 1% of lymphangiomas are found as pancreatic lesions and less than 100 cases are reported to date. Most lesions are asymptomatic, with no clear predominance in their location, and usually large, with reported diameters up to 20cm.1, 2 Based on the imaging findings, the differential diagnosis of a pancreatic cystic lymphangioma is broad and should exclude pseudocyst, cystadenoma, MCNs, IPMNs and cystic ductal carcinoma. CT shows a well-circumscribed, thin-walled, low-density, and homogenous cystic mass that may be unilocular or multilocular with thin-enhancing endocystic septae. These features are similar to cystadenomas that occur far more frequently. On MRI, the lesion appears hypodense on T1- sequence and hyperintense on T2. MRI is more useful to exclude communication between the cystic lesion and pancreatic duct when compared to CT.3 Some authors advocate the usefulness of EUS and EUS fine-needle aspiration (FNA) in the preoperative workup of large cystic lesions if a lymphangioma is suspected.3 In EUS imaging, a pancreatic lymphangioma usually appears as a well-defined, multi-cystic cavity with thin septae. In a recent review by Viscosi et. al, just a minority of cases performed EUS-FNA (6 out of 31), and this atitude has probably led to unnecessary surgical interventions.4 However, pancreatic lymphangiomas have a good prognosis when completely resected. A recurrence rate near 100% is expected if treated by aspiration or partial resection. Surgery should be considered in symptomatic patients with large cysts. To date, there are no reports of malignant transformation of pancreatic lymphangiomas.5 Lymphangiomas should be considered in the differential diagnosis of pancreatic cysts. EUS-FNA can give an appropriate diagnosis, preventing unnecessary surgical interventions in selected patients.
REFERENCES
- Carvalho D, Costa M, Russo P, et al. Cystic Pancreatic Lymphangioma – Diagnostic Role of Endoscopic Ultrasound. GE Port J Gastroenterol 2016; 23: 254-258. 2016/03/29. DOI: 10.1016/j.jpge.2016.01.006.
- Mohamed Sultan HP. Lymphangioma Presents as Peripancreatic Cystic Neoplasm: The Utility of Endoscopic Ultrasound. Journal of Clinical Trials 2014; 05. DOI: 10.4172/2167-0870.1000215.
- Fujii M, Saito H, Yoshioka M, et al. Rare Case of Pancreatic Cystic Lymphangioma. Intern Med 2018; 57: 813-817. 2017/11/21. DOI: 10.2169/internalmedicine.9445-17.
- Viscosi F, Fleres F, Mazzeo C, et al. Cystic lymphangioma of the pancreas: a hard diagnostic challenge between pancreatic cystic lesions-review of recent literature. Gland Surg 2018; 7: 487-492. 2018/12/07. DOI: 10.21037/gs.2018.04.02.
- Jayappa SN, Rao P, Tandon AS, et al. Large cystic lympangioma of the pancreas: a case reportum. Ann R Coll Surg Engl 2018; 100: e12-e14. 2017/10/20. DOI: 10.1308/rcsann.2017.0178.
AUTHORS
Filipe Taveira1, Luís Elvas1, Mafalda João1, Daniel Brito1, Ana Teresa Cadime1.
- Department of Gastroenterology, Portuguese Oncology Institute of Coimbra, Portugal.


