US Quiz of the Month – Fevereiro 2023
CASE REPORT
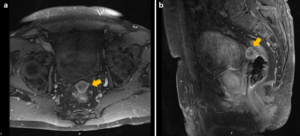
We present a case of a 75-year-old-male with medical history of an esophageal squamous cell carcinoma (cT4N0M0) who underwent radical chemoradiotherapy with external beam radiotherapy, paclitaxel and carboplatin. 6-month after finishing treatment, an abnormal pelvic fluorodeoxyglucose uptake (SUVmax 8.1) was seen in PET/CT (Fig. 1). MRI showed a 20*22mm heterogeneous nodular mass in the anterior peritoneal reflection, with contrast enhancement and diffusion restriction (Fig. 2). Thoraco-abdominopelvic CT showed no additional lesions.
Figure legends:
Fig. 1. PET/CT with abnormal pelvic fluorodeoxyglucose uptake (SUVmax 8.1), located anteriorly to the rectum.
Fig. 2. Axial (a) and sagittal (b) T1-weighted MRI with gadolinium revealing a 20*22mm heterogeneous nodular mass in the anterior peritoneal reflection, with contrast enhancement.
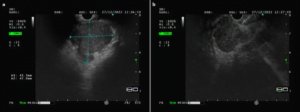
Fig. 3. Endoscopic ultrasound image showing a 41.1*42.6mm heterogeneous and predominantly hypoechoic lesion, with poorly defined contours (a), characterized by EUS-guided fine needle biopsy (b).
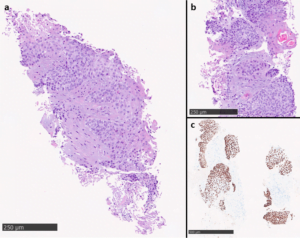
Fig. 4. Hematoxylin eosin (HE): biopsy sample composed of infiltrative neoplastic cells showing moderate cytologic atypia (a). Focal keratinization on HE slides (b) and nuclear p63 immunohistochemical expression in the neoplastic cells (c) support squamous differentiation.
What is the most likely diagnosis?
Endoscopic ultrasound (EUS) revealed a 41.1*42.6mm heterogeneous lesion, predominantly hypoechoic, with poorly defined contours, located in the anterior peritoneal reflection. The lesion had cleavage planes with adjacent pelvic structures. An EUS-guided fine needle biopsy of the lesion was performed using an 22G needle – 3 passes (Acquire™, Boston Scientific) (Fig. 3).
Histopathological evaluation showed a biopsy sample composed of multiple fragments of neoplasia and necrosis cells with moderate cytologic atypia, moderate eosinophilic cytoplasm and focal keratinization, invading the connective tissue. Diffuse nuclear p63 immunohistochemical expression was seen, supporting squamous differentiation (Fig.4). Considering histopathological findings and absence of other organ lesions on imaging studies, the diagnosis of peritoneal squamous cell carcinoma metastasis was made.
DISCUSSION
Peritoneal metastases (PM) generally arise from primary intraabdominal tumors (1). PM extra-abdominal origin is uncommon and true incidence is unknown. Breast and lung cancers are the most commonly extra-abdominal sites (2). Skin and urinary tract cancer are also common sources for PM (3). Peritoneal metastases from squamous cell esophageal cancer are extremely rare, with only one case reported in literature (4, 5). Due to its rarity, it is important to perform cross-sectional imaging studies (namely CT, MRI and PET/CT) to exclude origin for the metastases from other organs who can develop squamous cell carcinoma (breast, lung, skin, bladder).
REFERENCES
- Cortés-Guiral D, Hübner M, Alyami M, et al. Primary and metastatic peritoneal surface malignancies. Nat Rev Dis Primers. 2021;7(1):91.
- Flanagan M, Solon J, Chang KH, et al. Peritoneal metastases from extra-abdominal cancer – A population-based study. Eur J Surg Oncol. 2018;44(11):1811-1817.
- Rijken A, Galanos LJK, Burger JWA, et al. Peritoneal Metastases from Extraperitoneal Primary Tumors: Incidence, Treatment, and Survival from a Nationwide Database. Indian J Surg Oncol 2022.
- Ai D, Zhu H, Ren W, et al. Patterns of distant organ metastases in esophageal cancer: a population-based study. J Thorac Dis. 2017;9(9):3023-3030.
- Chen YH, Lin CY, Chen YT, et al. Unusual Metastases of Esophageal Squamous Cell Carcinoma. Clin Nucl Med. 2022;47(4):354-356.
AUTHORS
Renato Medas1,2, Pedro Moutinho-Ribeiro1,2, Sofia Pedrosa3, Pedro Canão2,3, Guilherme Macedo1,2
1 Gastroenterology Department, Centro Hospitalar Universitário de São João, Porto, Portugal
2 Faculty of Medicine of the University of Porto, Porto, Portugal
3 Pathology Department – Centro Hospitalar Universitário de São João